The
Electromagnetic Spectrum: the family of
|
|
Electromagnetic radiations are disturbances in an electric field.
|
![]() The Electromagnetic Spectrum is a continuous
band of electromagnetic radiation.
The Electromagnetic Spectrum is a continuous
band of electromagnetic radiation.
![]() It is not made up of particles of
matter - but rather of photons (a concept not met until A level - but the idea is that light is made up of photons which are little
quanta (or packets) of energy)).
It is not made up of particles of
matter - but rather of photons (a concept not met until A level - but the idea is that light is made up of photons which are little
quanta (or packets) of energy)).
![]() Electromagnetic radiation is composed
of rays of pure energy.All electromagnetic
rays have similarities in that they all travel at the same speed in
a vacuum - 3.0 x 108 m/s and are all made up of photons.
Electromagnetic radiation is composed
of rays of pure energy.All electromagnetic
rays have similarities in that they all travel at the same speed in
a vacuum - 3.0 x 108 m/s and are all made up of photons.
![]() Electromagnetic waves
differ in their wavelength, frequency, energy and method of production,
but their speed in a vaccuum is always the same.
Electromagnetic waves
differ in their wavelength, frequency, energy and method of production,
but their speed in a vaccuum is always the same.
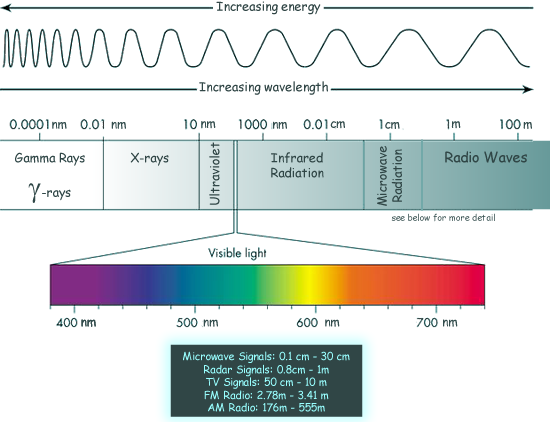
The parts of the electromagnetic spectrum are arranged in the table below in order of decreasing photon energy, decreasing frequency or increasing wavelength. Each part is named according to its origin and frequency/wavelength range. Light energy is the most familiar part of the spectrum and it is often referred to as the 'family of light'. Some parts of the e.m. spectrum can be directly detected by humans, others cannot. |
See the table below:
![]() indicates that the rays are harmful because they are of high enough energy
to be ionizing radiation.
indicates that the rays are harmful because they are of high enough energy
to be ionizing radiation.
Type of Electromagnetic
Energy |
Source of radiation |
Detectors
|
Typical Wavelength |
Uses |
Dangers |
|
|
Radioactive nuclei |
Geiger-Müller tube |
10-12 m |
Radiotherapy Diagnostic work with a gamma camera Pipe leakage |
|
|
|
X-ray tubes - produced when high energy electrons hit a metal target. Also given out from black holes and very bright stars |
Photographic film |
10-10 m (size of an atom) |
Medical scanning for broken bones and tumours in tissue |
cancer and mutations deep in the body |
|
|
Very hot objects, sun, sparks, mercury lamps. |
Photographic film, skin (it causes sun tans and skin cancer -see ozone layer depletion), makes fluorescent things glow with visible light. |
10-8 m |
Sterilisation |
skin cancer and cateracts |
Visible
Light |
Very
hot objects - like the Sun or stars, hot metals
(filament lamp), fluorescent objects, and visible lasers, LEDs.
|
Eyes,
photographic film, an LDR.
|
10-7 m (Red end is about 700nm - Blue end about 400nm) |
One of our main forms of one to one communication! Warning of dangers and location of food etc. Writing, painting etc. |
Very bright
light can damage the retina |
|
Warm or Hot objects, sun, IR lasers |
Skin, a blackened thermometer, a thermistor. |
10-5 m |
To keep us warm To cook food Remote control devices for TV etc. |
High intensity
can burn the skin and retina |
|
|
|
Short wave radio transmitters (cell phone communications) and microwave ovens (these contain a magnetron - a high-powered vacuum tube that generates coherent microwaves). . |
Aerial with a short wave radio set, or a satellite dish. Water filled objects (such as food) get very hot |
cm |
High intensity
and/or energy can cause heating effects within tissue - whether other
biological effects take place is debatable - some scientist think
it may cause tumour growth - but this is not by the route recognized
(it is none ionizing) |
|
| Radio and TV waves |
Radio and TV transmitters |
Aerial with a TV set or a radio set. (they CANNOT be heard!!!!!) |
km |
Media Communications |
None known
unless intensity is abnormally high |
![]() Most parts of the electromagnetic spectrum can be captured by telescopes to give us more information about the Universe.
Most parts of the electromagnetic spectrum can be captured by telescopes to give us more information about the Universe.
Follow me...