Atomic Structure
Q3.
(a) The most abundant isotope of cobalt is represented by 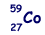 How many protons, neutrons and orbital electrons are there in a neutral atom of this element?
How many protons, neutrons and orbital electrons are there in a neutral atom of this element?
(2 marks)
(b) How is the nuclide that has one less proton than the nickel nuclide,  , represented?
, represented?
(2 marks)
(c)
(i) The heaviest isotope of hydrogen, whose nucleon number is 3, is called tritium. How is tritium represented?
(ii) Calculate the charge per unit mass, in C kg–1, for a tritium nucleus.
(3 marks)
(Total 7 marks)



