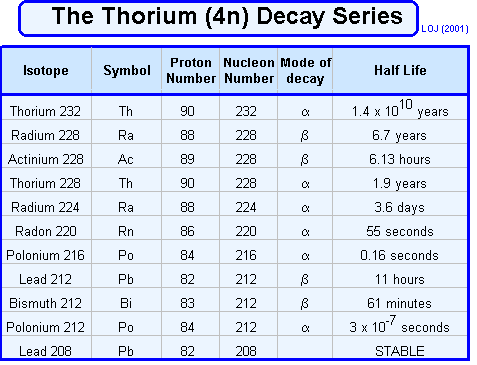
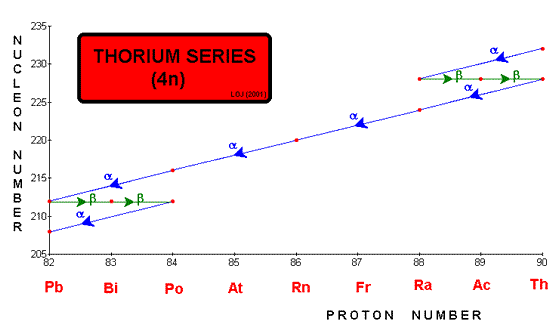
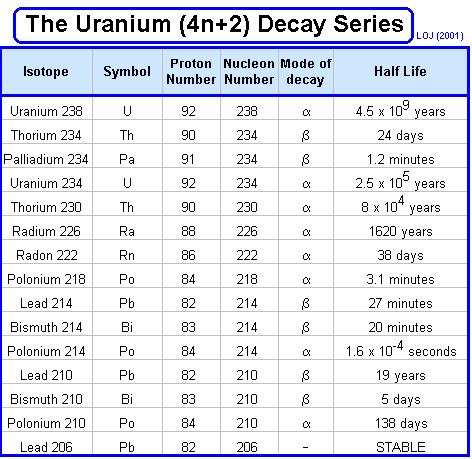
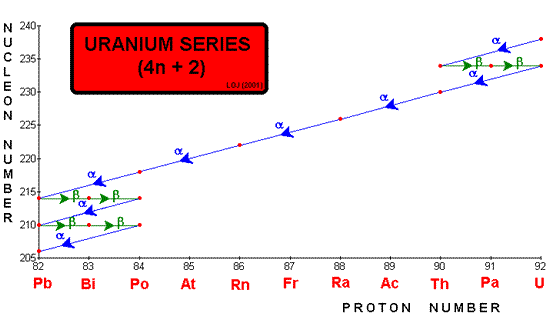
Radioactive series When ever a decay occurs the nucleon number either stays the same (gamma or beta emission) or reduces by four (alpha emission). Therefore if you divide the nucleon number by four you will either have no remainder or 1, 2 or 3. Mathematically we could say that the nucleon number could be described by 4n, 4n+1, 4n+2 or 4n+3. One unstable nucleus will decay into another unstable nucleus, over and over in a "decay series" until an ultra-stable nucleus (usually a stable isotope of lead Pb 206, Pb 207 or Pb 208) is reached as the end product. These are often represented graphically by plotting nucleon number against proton number.
Three such series occur naturally:
The
4n+3 series has only been possible since we made transuranic
elements in nuclear reactions.
|
Follow me...
|