Radioactive rocks
Uranium-238 has a very long half-life so deposits of it are still quite abundant around the world. It is an alpha particle emitter so it is not dangerous to living things when it is outside the body. However, tiny particles of rock are dissolved in water or broken off by the wind during the process of erosion. These tiny alpha emitters can be ingested (drank or eaten) or inhaled (breathed into the lungs). Alpha emitters inside the body are very
dangerous.
Igneous Rock: Granite
Sedimentary rock: limestone or sandstone Metamorphic rock
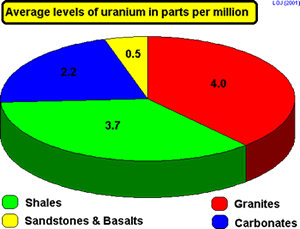
If a rock contains
a vein of uranium ore the concentration of uranium can be as great as
5000 parts per million but in general the levels are much lower.

Radioisotopes decay
to give other radioisotopes in a radioactive
decay series. Part of the decay series for uranium is the gas radon.
This means that rocks with uranium content are not only potentially
hazardous when tiny grains are transported by erosion but also from
the radon gas they emit. (See Radon Gas)
In recent geology carbon-dating can be used to date specimens as old as 35,000 years. |
Follow me...
|