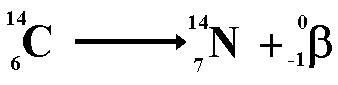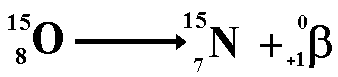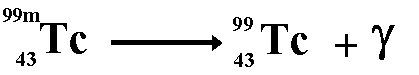Nuclear Equations
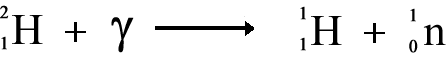
Neutron Decay
Alpha decayA nucleus that has high mass and too many protons to be stable tends to undergo alpha decay. When alpha decay occurs a group of two protons and two neutrons (helium nucleus) comes out of the nucleus. Therefore the proton number decreases by 2 but the nucleon number decreases by 4. The resulting daughter nucleus is of an element 2 positions to the left of the 'parent' in the periodic table.
The above equation
shows the radioactive decay of Uranium-238 by alpha
emission.
Therefore the nucleon numbers balance
Therefore the proton numbers balance
Beta DecayWhen a nucleus has too many neutrons, it tends to beta decay. When beta decay occurs a neutron within the nucleus emits the particle and changes into a proton. Therefore the proton number increases but the nucleon number stays the same (only now you have one more proton and one less neutron!). The resulting daughter nucleus is of an element 1 position to the right Beta decay is basically
the decay of a neutron into the more stable proton
Therefore the nucleon numbers balance
Therefore the proton numbers balance Positron DecayWhen a nucleus has too many protons, it tends to positron decay. A positron is an antimatter beta particle. When a positron meets with an electron it annihilates it! Both particles disappear and two gamma rays are produced instead. Positron decay is
the changing of a proton into a neutron.
The above equation shows the radioactive decay of Oxygen-15 by positron emission
Therefore the nucleon numbers balance
Therefore the proton numbers balance Gamma EmissionSometimes, after its emission of an alpha, beta or positron particle, the nucleus is still in an excited state, called a metastable state. In order to get to a lower energy state it emits a quantum of energy in the form of a gamma ray. This is not a highly unstable state otherwise the emission of the gamma ray would accompany the alpha, beta or positron particle. Nuclei in the metastable state produce gamma rays at a measurable half-life. E.g. Cobalt 60m decays
to give cobalt 60 with a half-life of 5.3 years and technetium 99m decays
to give technetium 99 with a half-life of 6 hours.
No matter is emitted from the nucleus therefore the nucleon number and the proton number remain the same. Before and after emission of the gamma ray they are the same isotope of the element but they are different nuclide because the term nuclide incorporates nuclear energy states as well basic structure. Neutron DecayIf a neutron rich nucleus is in an excited state - perhaps after bombardment with gamma rays - it will release a neutron to achieve a more stable one. When theis happens the number of protons does not change so we are still dealing with the same element and chemical properties will not change - but the neutron number does change - this changes the isotope we are dealing with and the physical properties of the element.
|
Follow me...
|


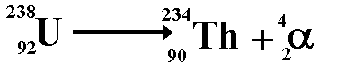
 Look at the
numbers on the top line (the nucleon numbers).
Look at the
numbers on the top line (the nucleon numbers).