A Level Specific Heat Capacity Questions
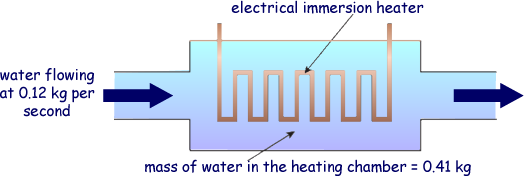
Q3. An electrical immersion heater supplies 8.5 kJ of energy every second. Water flows through the heater at a rate of 0.12 kg s–1 as shown in the diagram.
(a) Assuming all the energy is transferred to the water, calculate the rise in temperature of
the water (in kelvin) as it flows through the heater, given that the
specific heat capacity of water = 4200 J kg–1K–1
(2 marks)
(b) The water suddenly stops flowing at the instant when its average temperature is 26o C. The mass of water trapped in the heater is 0.41 kg. Calculate the number of seconds it takes for the water to reach 100o C if the immersion heater continues supplying energy at the same rate.
(2 marks)
(Total 4 marks)








