A Level Specific Heat Capacity Questions
Q2. An electrical heater is used to heat a 1.0 kg block of metal, which is well lagged. The table shows how the temperature of the block increased with time.
temperature/°C |
20.1 |
23.0 |
26.9 |
30.0 |
33.1 |
36.9 |
time/s |
0 |
60 |
120 |
180 |
240 |
300 |
(a) Plot a graph of temperature against time
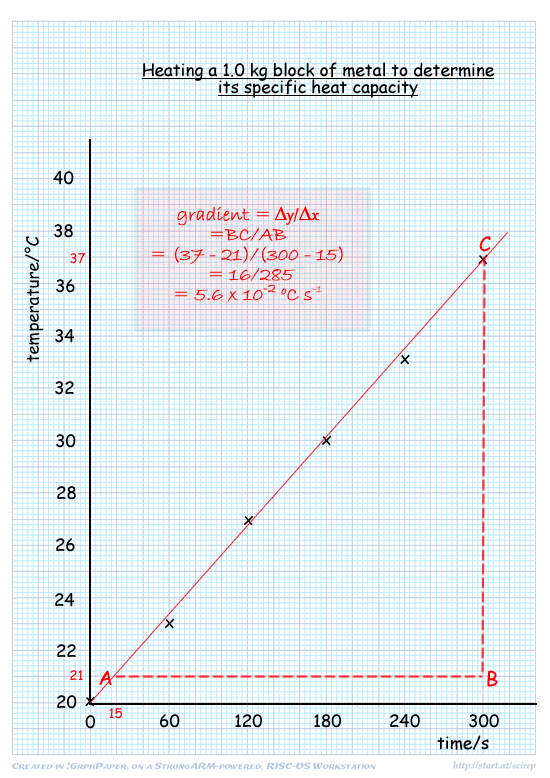
- adequate scale filling as much of the graph paper as possible- neither x nor y scale should be able to be doubled and still aloow plotting of the points on the graph paper

- points plotted correctly

- best fit line (at least one point to right and left of line - but ideally two each side)

(3 marks)
(b) Determine the gradient of the graph.
Use of triangle with sides that are of at least half the size of each axis line 
gradient = 0.056 ± 0.004 (°C/s) 
(2 marks)
(c) The heater provides thermal energy at the rate of 48 W. Use your value for the gradient of the graph to determine a value for the specific heat capacity of the metal in the block.
P = Q/t
P = mc T/t = mc x (gradient of graph)
T/t = mc x (gradient of graph)
48 = 1.0 x c x 0.056 
c = 48/0.056
c = 860 J kg–1 K–1 (or J kg–1 °C–1) 
(2 marks)
(d) The heater in part (c) is placed in some crushed ice that has been placed in a funnel as shown.
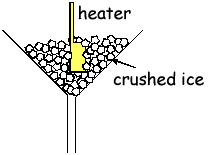
The heater is switched on for 200s and 32g of ice are found to have melted during this time. Use this information to calculate a value for the specific latent heat of fusion for water, stating one assumption made.
Energy supplied by the heater in 200s = 48 x 200 = 9600 J 
This energy is used to melt the ice:
9600 = ml
l = 9600/(32 x 10-3)
= 3.0 x 105 J kg–1 
Any sensible assumption can be stated:
no heat lost to surroundings
OR
temperature
does not change
OR
all heat is transferred to ice 
(3 marks)
(Total 10 marks)








