Rutherford Scattering - A Level Standard Questions
Q2. In an experiment to investigate the structure of the atom, alpha particles were aimed at thin gold foil in a vacuum. A detector was used to determine the number of alpha particles deflected through different angles.
(a)
(i) In which direction will the number of a particles per second be a maximum?
Straight on (or deflection of zero degrees)
(ii) State what this result suggests about the structure of the atoms in the metal.
The atom consists mainly of empty space [or the volume of the nucleus is (very much) smaller than volume of the atom] 
(2 marks)
(b) A small number of alpha particles are scattered through 180°. Explain what this suggests about the structure of the atoms in the metal.
It suggests that
- most of the mass of an atom is contained in its nucleus [or the mass of the nucleus is greater than the mass of the alpha particle]

- the nucleus contains a positive charge

- the charge is concentrated at the nucleus

(3 marks)
(c) The figure shows the path of an alpha particle passing near a nucleus.
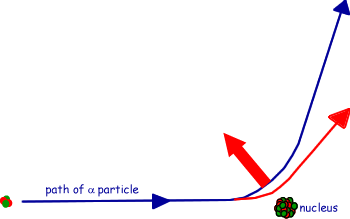
(i) Name the force that is responsible for the deflection of the alpha particle.
The electrostatic force or electromagnetic force or coulomb force 
(ii) Draw an arrow on the diagram in the direction of the force on the alpha particle in the position where the force is a maximum.
See diagram
- arrow pointing away from the nucleus at the closest distance to the nucleus 
(iii) The nucleus is replaced with one which has a larger mass number and a smaller proton number. Draw on the diagram the path of an alpha particle that starts with the same velocity and position as that of the alpha particle drawn.
See diagram - path showing less deflection at all times 
(3 marks)
(Total 8 marks)







