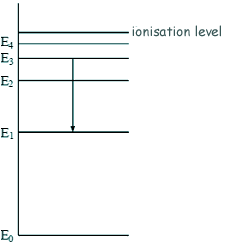
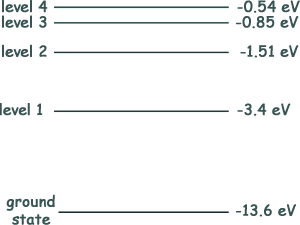Energy Transistions - Multiple Choice Q1.The diagram shows some energy levels of an atom.
The transition E3 to E1 corresponds to the emission of visible light. A transition corresponding to the emission of infrared radiation could be:
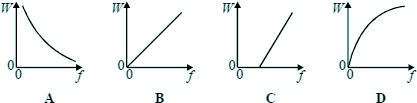
Q2. Which one of the graphs below best represents the relationship between the energy W of a photon and the frequency f of the radiation?
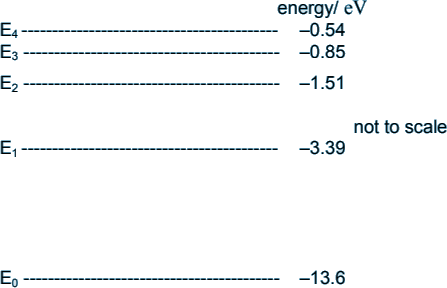
Q3. The diagram shows some of the energy levels for a hydrogen atom.
A free electron of kinetic energy 20.0 × 10–19 J collides with a hydrogen atom in its ground state. The hydrogen atom is excited from its ground state to the first excited state. Calculate the kinetic energy of the free electron after the collision in joules.
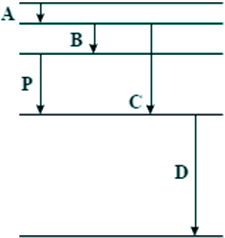
Q4. The diagram below is drawn to scale. It shows some of the energy levels of an atom.
Transition P results in the emission of a photon of wavelength 4 × 10–7 m. Which one of the transitions A, B, C, or D could result in the emission of a photon of wavelength 8 × 10–7 m?
Q5. The diagram gives some of the energy levels of a hydrogen atom.
The transition of an excited hydrogen atom from E3 to E1 causes a photon of visible light to be emitted. Which transition causes a photon of ultraviolet light to be emitted?
Q6. The diagram below shows an energy level diagram for a hydrogen atom.
Electrons with energy 13.0 eV collide with atoms of hydrogen in their ground state. What is the number of different wavelengths of electromagnetic radiation that could be emitted when the atoms de-excite?
Q7. The diagram shows an energy-level diagram for a hydrogen atom.
Electrons, each having a kinetic energy of 2.0 × 10−18 J, collide with atoms of hydrogen in their ground state. Photons are emitted when the atoms de-excite. How many different wavelengths can be observed with incident electrons of this energy?
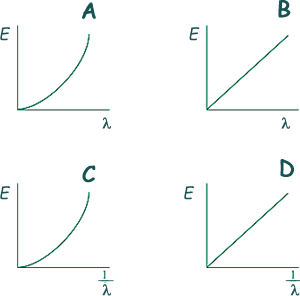
Q8. Which graph best shows the relationship between photon energy E and wavelength of a photon of electromagnetic radiation?
|
Follow me...
|