GCSE Questions: Radioactivity
Q7.
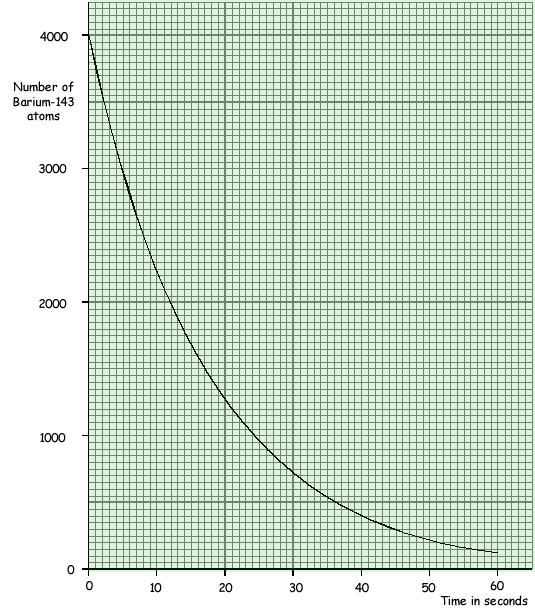
(a) The graph shows how a sample of barium-143, a radioactive isotope with a short half-life, decays with time.
(i) What is meant by the term isotope?
Isotopes of an element have equal number of protons but different number of neutrons 

Isotopes of an element have the same atomic/proton number but a different mass/nucleon number 
(1 mark)
(ii) What is meant by the term half-life?
Half-life is the time taken for activity (or 'count rate' or 'number of nuclei') to decrease to half. 

(1 mark)
(iii) Use the graph to find the half-life of barium-143.
12 seconds 
(1 mark)
(b) Humans take in the radioactive isotope carbon-14 from their food. After their death, the proportion of carbon-14 in their bones can be used to tell how long it is since they died. Carbon-14 has a half-life of 5700 years.
(i) A bone in a living human contains 80 units of carbon-14. An identical bone taken from a skeleton found in an ancient burial ground contains 5 units of carbon-14.
 Calculate the age of the skeleton.
Calculate the age of the skeleton.
 Show clearly how you work out your answer.
Show clearly how you work out your answer.
80 to 40 - First half life
40 to 20 - Second half life
20 to 10 - Third half life
10 to 5 - Fourth half life
4 half lives  = 4 x 5700 = 22800 years
= 4 x 5700 = 22800 years 
(2 marks)
(ii) Why is carbon-14 unsuitable for dating a skeleton believed to be about 150 years old?
The decay of carbon-14 over 150 years is insignificant  there is very little decay
there is very little decay  the change is too small
the change is too small 
(1 mark)
(c) The increased industrial use of radioactive materials is leading to increased amounts of radioactive waste.
Some people suggest that radioactive liquid waste can be mixed with water and then safely dumped at sea.
 Do you agree with this suggestion?
Do you agree with this suggestion?
 Explain the reason for your answer.
Explain the reason for your answer.

FOR
 massive dilution of waste within the ocean
massive dilution of waste within the ocean
 reduces concentration (within a given volume) to insignificant levels
reduces concentration (within a given volume) to insignificant levels 
 dumped distant from human habitation
dumped distant from human habitation 
AGAINST
 pollution (of the sea/beach)
pollution (of the sea/beach) 
 mutation or harm caused to living things (animals/plants)
mutation or harm caused to living things (animals/plants) 
 effect on food chain
effect on food chain 
 long period of time necessary for radioactive isotopes to be harmless
long period of time necessary for radioactive isotopes to be harmless 
(3 marks)
(Total 9 marks)


