GCSE Questions: Nuclear Structure
Q1. The diagram below shows the paths of two alpha particles A and B into and out of a thin piece of metal foil.
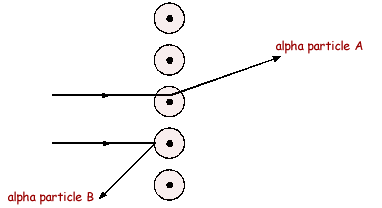
(a) The paths of the alpha particles depend on the forces on them in the metal. Describe the model of the atom which is used to explain the paths of alpha particles aimed at thin sheets of metal foil.
(3 marks)
(b) Scientists used to believe that atoms were made up of negative charges embedded in a positive 'dough'. This is called the 'plum pudding' model of the atom.
The diagram below shows a model of such an atom.
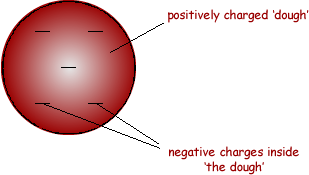
(i) Explain how the 'plum pudding' model of the atom can explain why alpha particle A is deflected through a very small angle.
(2 marks)
(ii) Explain why the 'plum pudding' model of the atom can not explain the large deflection of alpha particle B.
(3 marks)
(c) We now believe that atoms are made up of three types of particles called protons, neutrons and electrons.
Complete the table below to show the relative mass and charge of a neutron and an electron.
The relative mass and charge of a proton have already been done for you.
PARTICLE |
RELATIVE MASS |
RELATIVE CHARGE |
proton |
1 |
+1 |
neutron |
|
|
electron |
|
|
(2 marks)
(d) The diagrams below show the nuclei of four different atoms A, B, C and D.
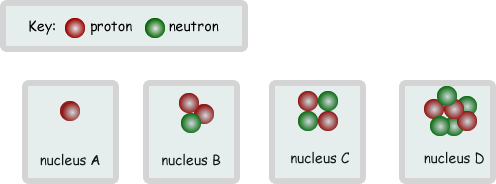
(i) State the mass number of C.
(ii) Which two are isotopes of the same element? Explain your answer.
(4 marks)
(Total 14 marks)










