Solutions: Radioactivity Questions
Q12. The isotope of uranium,  , decays into a stable isotope of lead,
, decays into a stable isotope of lead,  by means of a series of α and β– decays.
by means of a series of α and β– decays.
(a) In this series of decays, α decay occurs 8 times and β– decay occurs n times. Calculate n.
92 - 16 = 76 and
The final proton number is 82 - there are therefore 82 - 76
= 6 beta decays.
(1 mark)
(b)
(i) Explain what is meant by the binding energy of a nucleus.
The binding energy of a nucleus is the energy required to split the nucleus up  into its individual neutrons and protons (or nucleons)nucleons
into its individual neutrons and protons (or nucleons)nucleons 
OR the energy released to form the nuleus  from its individual neutrons and protons/nucleons
from its individual neutrons and protons/nucleons 
(2 marks)
(ii) The graph belowshows the binding energy per nucleon for some stable nuclides.
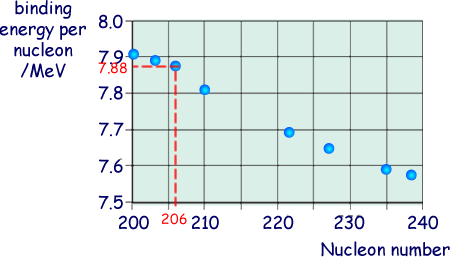
Estimate the binding energy, in MeV, of the  nucleus.
nucleus.
From the graph the binding energy per nucleon is 7.88 MeV
We have 206 nucleons, so the total binding energy is 206 x 7.88 = 1620 MeV 
(1 mark)
(c) The half-life of  is 4.5 × 109 years, which is much larger than all the other half-lives of the decays in the series. A rock sample when formed originally contained 3.0 × 1022 atoms of
is 4.5 × 109 years, which is much larger than all the other half-lives of the decays in the series. A rock sample when formed originally contained 3.0 × 1022 atoms of  and no
and no  atoms.
atoms.
At any given time most of the atoms are either  or
or  with a negligible number of atoms in other forms in the decay series.
with a negligible number of atoms in other forms in the decay series.
(i) Using the axes below, sketch graphs to show how the number of  atoms and the number of
atoms and the number of  atoms in the rock sample vary over a period of 1.0 × 1010 years from its formation.
atoms in the rock sample vary over a period of 1.0 × 1010 years from its formation.
Label your graphs U and Pb.
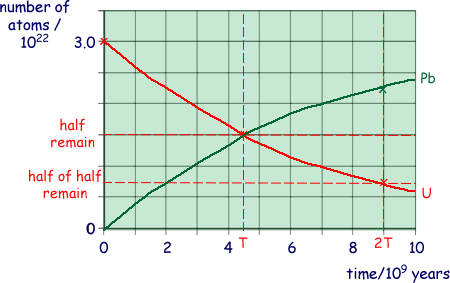
U, a graph starting at 3.0 × 1022 showing exponential fall passing through 0.75 × 1022 at 9 × 109 years 
Pb, inverted graph of the above so that the graphs cross at 1.5 × 1022 at 4.5 × 109 years 
(2 marks)
(ii) A certain time, t, after its formation the sample contained twice as many  atoms as
atoms as  atoms.
atoms.
Show that the number of  atoms in the rock sample at time t was 2.0 × 1022.
atoms in the rock sample at time t was 2.0 × 1022.
Let number of uranium atoms be U and number of lead atoms be Pb
At time t
U + Pb = 3.0 × 1022
But U = 2Pb
So, U + 0.5U = 3.0 × 1022
U = 3.0 × 1022/1.5 = 2.0 × 1022
(1 mark)
(iii) Calculate t in years.
N = No e-λt
2 × 1022 = 3 × 1022 × e-λt
ln (2/3) =
-λt
ln (3/2) = ln 1.5 = λt
t = ln 1.5 / λ 
Now λ = ln 2 / t1/2
λ = ln 2 / 4.5 × 109
λ = 1.54 × 10-10 
t = ln 1.5/ 1.54 × 10-10
t = 2.6 × 109 years 
(3 marks)
(Total 10 marks)


