Solutions: Atomic Structure
Q12.
(a) State what is meant by the specific charge of a nucleus and give an appropriate unit for this quantity.
Specific charge is the ratio of charge to mass of the nucleus 
The unit is C kg–1 
(2 marks)
(b) Nucleus X has the same nucleon number as nucleus Y. The specific charge of X is 1.25 times greater than that of Y.
(i) Explain, in terms of protons and neutrons, why the specific charge of X is greater than that of Y.
The number of protons plus the number of neutrons is the same, therefore the mass is virtually the same for both nuclei  but if X has more protons (or less neutrons) than Y is will have a greater charge and therefore the specific charge will be greater.
but if X has more protons (or less neutrons) than Y is will have a greater charge and therefore the specific charge will be greater. 
(2 marks)
(ii) Nucleus X is 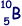 . Deduce the number of protons and the number of neutrons in nucleus Y.
. Deduce the number of protons and the number of neutrons in nucleus Y.
Let 'm' be the mass of a proton or neutron.
Specific charge of X is 5e/10m = 0.5 e/m
Specific charge of Y is 0.5/1.25 x e/m = 0.4 e/m = 4e/10m
Therefore there are 4 protons  in the nucleus and 6 neutrons.
in the nucleus and 6 neutrons.
Answer: 6 neutrons 
(4 marks)
(Total 8 marks)


