| 3.2.2.1 The photoelectric effect |
Threshold frequency; photon explanation of threshold frequency.
Work function Φ
stopping potential Vs
threshold frequency fo
EKmax is the maximum kinetic energy of the photoelectrons.
photoelectric equation hf = Φ + EKmax
(the stopping potential experiment is not required) |

(QUESTIONS 1-4: L&R Ex 27.1) |
- Explain the underlying principles behind the photoelectric effect, including a derivation of Einstein’s equation in terms of energy conservation
- Understand the term photon and calculate the number of photons in a light beam
- Derive the equation: hf = Φ + EKmax
- Explain that the work function is the energy with which each electron is bound to the surface of the metal (and therefore the energy that is required to release it from the metal)
You will have seen a demonstration of the photoelectric effect using a photocell or an electroscope with a zinc plate attachment and UV lamp.
|
| 3.2.2.2 Collisions of electrons with atoms: |
Ionisation, excitation
Understanding of the role of ionisation and excitation in the fluorescent tube;
The electronvolt
Students will be expected to be able to convert eV into J and vice versa.
|
|
- Recall the structure of the atom to include the energy level model for electrons
- Recall the definition of the electronvolt |
| 3.2.2.3 Energy levels and photon emission |
Line spectra (e.g. of atomic hydrogen) as evidence of transitions between discrete energy levels
Energy levels, photon emission :
hf = E1 - E2
In questions, energy levels may be quoted in J or eV |

Ciccotti and Kelly pp 72-79
L&R Ex136 Q1, 2, 5, 7
(QUESTION 7: L&R Ex 27.1)
Muncaster G19-G21 |
- There are two types of line spectra: absorption and emission
- Explain the difference between ionisation and excitation and apply this to the operation of a fluorescent tube
- Explain the origin of line spectra and use this as evidence to support the model of discrete energy levels
- Recall the equation hf = E1 - E2
You will have made an observation of line spectra using a diffraction grating. |
3.2.2.4
Wave-particle duality |
Students should know that electron diffraction suggests that particles possess wave properties and the photoelectric effect suggests that electromagnetic waves have a particulate nature. Details of particular methods of particle diffraction are not expected.
de Broglie wavelength
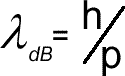
Students should be able to explain how and why the amount of diffraction changes when the momentum of the particle is changed.
Appreciation of how knowledge and understanding of the nature of matter changes over time.
Appreciation that such changes need to be evaluated through peer review and validated by the scientific community |

Ciccotti and Kelly pp 83
(QUESTIONS 5-6: L&R Ex 27.1)
|
- Recall the evidence for wave and particle behaviour in light and matter and indicate practical methods that can be used to demonstrate these properties
- Recall the equation for the de Broglie wavelength
- Recognise the effect of particle speed on wavelength and the potential diffraction effect that could be obtained
You will have seen a demonstration using an electron diffraction tube.
You should be able to use prefixes when expressing wavelength values.
|


